Interleukin-1 Alpha Polymorphisms during Chemotherapy in Breast Cancer Patients
Article Type
Research Article
Online Date
March 05, 2025
Open Access
Yes
Ahmed T.H. Faris Al-Karkhi*
Department of Biology, College of Medicine, Al-Mustaqbal University, Babylon, Iraq
*Corresponding author:, Ahmed T.H. Faris Al-Karkhi, College of Medicine, Al-Mustaqbal University, 51001 Babylon, Iraq
Citation Al-Karkhi ATHF (2025) Interleukin-1 Alpha Polymorphisms during Chemotherapy in Breast Cancer Patients Glob J Surg Surgical Res 1(2): 70-74.
Received Date: 21 May 2025; Accepted Date: 22 July 2025; Published Date: 01 August 2025
Abstract
Objective: The IL-1 alpha -889 C>T polymorphism in the promoter region of IL-1 alpha has been associated with an increased risk of breast cancer.
Method: In 100 breast cancer patients, 70 tissue samples, and 50 control samples, the genotype frequencies of the IL-1 alpha -889 C>T promoter polymorphism were examined using RFLP and PCR.
Result: In the RFLP-PCR allele frequencies of IL-1 alpha -889 C>T in both healthy individuals and patients, the CT allele had a greater effect on leukemia patients than the TT allele (OR 1.28, 0.72). However, in patients with tissue-related tumors, the effect of the CT allele frequency was greater than that of the TT allele (1.00 vs. 0.33), while the opposite was true in patients with both malignant and benign tumors: the TT allele frequency had a greater effect than the CT allele (0.90 vs. 0.70).
Conclusion: According to the IL1 alpha -889 C>T gene score, the T allele was a risk factor in the blood at p = 0.002*, and the TT genotype showed a greater effect in the blood at p = 0.007*, with statistical significance only between patients with blood and tissue-related tumors.
Keywords: Breast Cancer, IL1 α; Polymorphism
Introduction
The diagnosis of soft tissue lesions of the breast is often challenging in routine practice. They can be roughly classified as benign or malignant tumors and encompass a wide range of histopathological abnormalities. Breast cancer is the most common cancer among women [1]. The majority of breast lesions are found in females [2]. BRCA1/2, a nuclear phosphoprotein associated with DNA repair, is involved in both the cell cycle and the response to DNA damage [3]. Immune and inflammatory responses are initiated and regulated by the Interleukin-1 (IL-1) family of cytokines. [4] Both members of the IL-1 family exhibit similar biological activity, despite being encoded by different genes. This results in the duplication of IL-1R1 with itself [5]. Although epithelial cells and other cell types regularly express IL-1α under steady-state conditions, pro-inflammatory and stress-related events lead to its elevated expression. There are three ways in which this cytokine acts intracellularly [6]. Third, similar to transcription factors, IL-1α can translocate from the cytosol to the cell nucleus and perform intracellular functions [7-19]. Both sterile and pathogen-induced inflammation are initiated by IL-1α and T helper 17 (Th17) responses [4,11].
IL-1α is a transcription factor located in the nucleus and regulates both normal cell growth and tumorigenesis. IL-1α acts as a “prognostic factor” [12-14], when cells die through processes such as necrosis. Images from breast ultrasound. Morphological and histological features are often used to differentiate between cancerous and benign tumors. A straightforward approach relies on highly skilled radiologists with extensive experience manually evaluating images for texture and morphological features to determine whether a tumor is benign or malignant [15-17] and classifying tumors as benign or malignant using classifiers based on morphological and textural features [18]. Benign breast changes are more common in women of reproductive age, peaking between the ages of 30 and 50. Conversely, during the postmenopausal phase [19]. Monitoring, counseling, and symptomatic relief are part of the management of benign breast changes in those at high risk for breast cancer. Histological, clinical, and radiographic examinations are used in diagnostic methods to exclude cancer. A comprehensive history and clinical evaluation are required in conjunction with diagnostic imaging directed at these symptoms. Fibrocystic breast changes, a very common benign disease affecting up to 50% of premenopausal women, are also referred to as fibrocystic mastopathy [20]. Fibrocystic mastopathy is associated with histopathological changes [21-23]. The polymorphisms of the IL1 alpha gene in the blood and tissues of breast cancer patients is the aim of this research.
Materials
A patient from BC provided 100 blood samples before starting treatment. The IL-1 alpha polymorphism -889 C>T was found in 50 healthy women's blood samples using DNA from both patients' and healthy people's whole blood. Between September 2021 and October 2022, women aged 14-66 who worked at AL-Hilla-Teaching Hospital, Imam Sadiq Hospital, and Marjan Hospital were included in the study.
IL-1 alpha -889 C>T Genotyping
As directed by the manufacturer, Magna Pure LC (Roche, Germany) was used to extract genomic DNA from 2 milliliters of peripheral blood leukocytes. PCR-RFLP (Restriction Fragment Length Polymorphism) was used to determine each genotype. Using the forward primer (5′-TTACATATGAGCCTTC-CATG-3?) and the reverse primer (5′-AAGCTTGTTCTACCACCT-GAACTAGGC-3? ((17))), the IL-1a -889 C>T promoter polymorphism was identified. For the amplification of DNA samples from each patient and control group, 200 ng of DNA and 2.5 mmol/l Magnesium Chloride (MgCl) were added to a 50 μl reaction volume. Among the reagents utilized were Taq DNA polymerase (2.5 U/μl), oligonucleotide primers (10 pmol each), and Deoxyribonucleotide Triphosphates (dNTPs). An initial denaturation step at 94°C for five minutes preceded each of the 35 cycles of the PCR process, which included one minute at 94°C, one minute at 53.9°C, and two minutes at 72°C for extension. At 72°C, a last extension was performed for 5 minutes. Once the 108-bp PCR product was combined with the Nco I restriction enzyme, it was left overnight at 37°C. To differentiate the genotype, 25 µL of the digested sample was stained with ethidium bromide and then run through 3% agarose gel electrophoresis
Results
IL-1 alpha -889 C>T Promoter primer
A specific primer was used to produce the PCR result for the IL-1 alpha -889 C>T gene. The PCR result (band) for blood, tissue, and healthy individuals was 108 bp (Figures1&2).
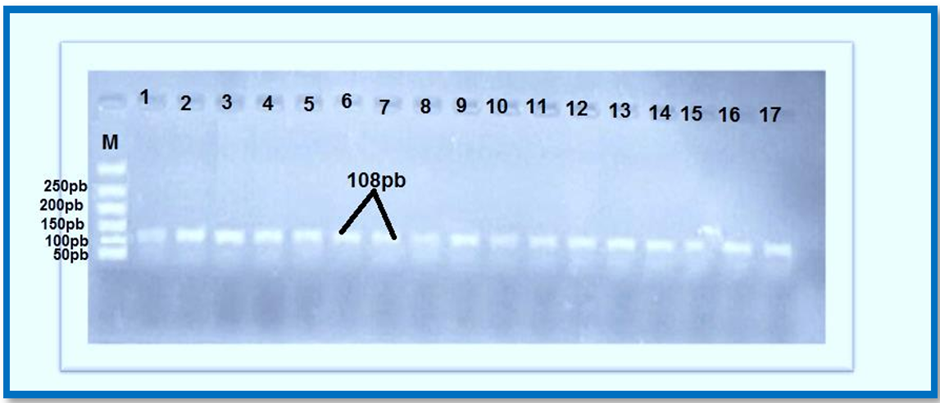
Figure 1: IL-1 alpha -889 C>T, M: molecular DNA ladder, 1-17 pattern of PCR product electrophoreses 53.9, 85 volts, and one hour was the optimal annealing temperature.

Figure 2: M: DNA ladder and PCR product (108 bp) showing the electrophoresis pattern of the IL-1 alpha -889 C>T gene PCR-RFLP on a PAGE gel using the restriction enzyme NocI. The TT homozygote genotype (92 bp) is observed in lanes 5, 9, and 10; the CC homozygote genotype (108 bp) appears in lanes 2, 3, and 8; while the heterozygote CT genotype (both 108 bp and 92 bp) is visible in lanes 1, 4, 6, 7, and 11. Electrophoresis was performed at 100 volts for one hour.
IL-1 alpha-889 genotype frequency detection Using PCR-RFLP, the C>T gene polymorphism linked to breast cancer (Tables 1-3)
|
Genotype IL-1g alpha -889 C>T |
Blood patients |
Healthy (control) |
P- value |
Odd ratio |
|
CC |
27(27%) |
11(23%) |
||
|
CT |
22(30%) |
27(40%) |
0.51 |
1.44 |
|
TT |
43(43%) |
16(34%) |
0.33 |
0.58 |
|
Total number |
100 |
50 |
||
|
Allele frequency |
||||
|
C |
84 (44%) |
46 (45%) |
||
|
T |
16 (59%) |
44 (41%) |
0.36 |
0.78 |
Table 1: The allele and genotype frequencies of the IL-1 alpha-889 C>T gene polymorphism was analyzed in patients with blood-related breast cancers and compared to healthy controls.
|
Genotype IL-1 alpha -889 C>T |
blood patients |
Tissue patients |
P- value |
Odd ratio |
|
CC |
24(27%) |
11(23%) |
||
|
CT |
22(30%) |
27(40%) |
0.01 |
22(30%) |
|
TT |
45(43%) |
16(34%) |
0.002 |
43(43%) |
|
Total number |
100 |
81 |
100 |
|
|
Allele frequency |
||||
|
C |
77 (44%) |
86 (56%) |
||
|
T |
16 (59%) |
11 (41%) |
0.007 |
21 (59%) |
Table 2: Allele and genotype frequencies of the IL-1 alpha-889 C>T gene polymorphism have been studied in patients diagnosed with breast tissue cancers and those with blood-related conditions.
|
OR CL 95% |
P value |
Benign Blood |
Malignant Blood |
Genotype IL-1 alpha -889 C>T |
|
20(34%) |
12(29%) |
CC |
||
|
0.70(0.28-1.76) |
0.46 |
26 (45%) |
22(52%) |
CT |
|
0.90( 0.28-2.83) |
0.85 |
12 (21%) |
8 (19%) |
TT |
|
58 |
42 |
Total |
||
|
Alleles |
||||
|
66(0.55) |
46(0.55) |
C |
||
|
0.91( 0.52-1.61) |
0.76 |
50(0.45) |
38(0.45) |
T |
Table 3: Genotype and allele frequencies of the IL-1 alpha-889 C>T gene polymorphism in patients with benign and malignant blood and breast cancers.
Table 2 presents the allele frequencies of IL-1 alpha -889 C>T observed in the blood and tissue samples of patient participants. According to the Hardy-Weinberg equation, the T and C allele frequencies were 77 (0.44) and 86 (0.56) in blood samples, and 16 (0.59) and 11 (0.41) in tissue samples, respectively. Table 3 presents the frequencies of the IL-1 alpha -889 C>T allele in the blood of patients with both benign and malignant conditions. Using the Hardy-Weinberg equation, the allele frequencies for T and C were determined to be 46 (0.55) and 66(0.55) in patients with benign blood, and 38(0.45) and 46(0.55) in those with malignant blood.
Discussion
The genotype frequencies of the IL-1 alpha -889 C>T gene polymorphism in healthy individuals were 27 (27%) for TT, 22 (30%) for CT, and 11 (23%) for CC. In contrast, in blood patients, these frequencies were 43, 45, and 8, respectively (Table 1). Genotype CT patients experienced breast tumors roughly once more often than those with genotype TT (odd ratio = 1.20 and 0.77). The gene frequencies of IL-1 alpha -889 C>T were found to be non-significant, with P-values of 0.46 for CT and 0.76 for TT. Based on the data in Table 2, among patients with breast cancer, the distribution of the IL-1 alpha -889 C>T gene polymorphism genotypes was as follows: TT in 15 patients (24%), CT in 30 patients (49%), and CC in 27 patients (33%). The frequencies of these genotypes revealed that TT showed a statistically significant P-value of 0.001 (p<0.05), whereas the CT and CC genotypes did not exhibit significance. Additionally, the prevalence of breast tumors was similar between individuals with the CT genotype and those with the TT genotype, with odds ratios of 1.00 and 0.33, respectively. The frequencies of the TT, CT, and CC genotypes for the IL-1 alpha-889 C>T gene polymorphism were 12 (21%), 26g (45%), and 20 (34%) respectively. In blood-bent individuals, these frequencies were 8 (19%), 22 (52%), and 12 (29%). As shown in Table 3, the P-values for the genotype frequencies of the IL-1 alpha -889 C>T gene indicate that patients with breast tumors who had the TT genotype were more likely to have breast tumors compared to those with the CT genotype (Odds ratio 0.90, 0.70).
Conclusion
The IL-1 alpha-889 C>T gene polymorphism genotype frequencies in tissue-beneathless patients were 9 (33%) for TT, 15 (19%) for CT, and 22 (48%) for CC. Table 3 shows that these frequencies were 5 (21%), 7 (29%), and 12 (50%), respectively, in malignant individuals. Even though patients with the CT genotype were more likely to have breast cancers than those with the TT genotype (Odds ratio 1.16, 0.98), the P-value for the genotype frequencies of the IL-1 alpha -889 C>T gene in patients with breast malignancies did not reveal any substantial difference. Tissue allele frequencies for IL-1 alpha -889 C>T in both malignant and benign patient groups are shown in Table 3. The Hardy-Weinberg equation showed that the frequencies of the T and C alleles were 17(0.35) and 31(0.66) in patients with malignant blood, and 33(0.36) and 59(0.64) in individuals with benign blood.
References
- Wang, J, Shi, Y, Wang G, Dong S, et al. (2019) The association between interleukin?1 polymorphisms and their protein expression in Chinese Han patients with breast cancer. Molecular Genetics & Genomic Medicine 7: 804.
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, et al. (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clinic 71: 209-249.
- Hatim KS, Laxmikant NS, Mulla T (2017) Patterns and Prevalence of Benign Breast Disease in Western India. International Journal of Research in Medical Sciences 5: 684-688.
- Savaridas SL, Taylor DB, Gunawardana D, Phillips M (2017) Could parenchymal enhancement on contrast-enhanced spectral mammography (CESM) represent a new breast cancer risk factor? Correlation with known radiology risk factors Clinical Radiology 72: 1085.
- McCann B, Miaskowski C, Koetters T, Baggott C, West C, et al. (2012) Associations between pro-and anti-inflammatory cytokine genes and breast pain in women prior to breast cancer surgery. The journal of pain 13: 425-437.
- Garlanda C, Dinarello CA, Mantovani A (2013) The Interleukin-1 Family: Back to the Future. Immunity 39: 1003-1018.
- Merriman JD, Aouizerat BE, Cataldo JK, Dunn L, Cooper BA et al. (2014) Association between an interleukin 1 receptor, type I promoter polymorphism and self-reported attentional function in women with breast cancer. Cytokine 65: 192-201.
- Verstrepen L, Bekaert T, Chau TL, Tavernier J, Chariot A, et al. (2008) TLR-4, IL-1R and TNF-R signaling to NF-kappaB: Variations on a common theme. Cell. Mol. Life Sci 65: 2964-2978.
- Cohen I,Rider P, Carmi Y, et al. (2010) Differential release of chromatin-bound IL-1alpha discriminates between necrotic and apoptotic cell death by the ability to induce sterile inflammation, Proc Natl Acad Sci USA 107: 2574-2579.
- Liu F, Li L, Lan M, Zou T, Kong Z, et al. (2021) Key Factor Regulating Inflammatory Microenvironment, Metastasis, and Resistance in Breast Cancer: Interleukin?1 Signaling. Mediators of Inflammation 7785890.
- Netea MG, van de Veerdonk FL, van der Meer JW, Dinarello CA, Joosten LA (2015) Inflammasome-independent regulation of IL-1-family cytokines. Rev Immunol 33: 49-77.
- Chung Y, Chang SH, Martinez GJ, Yang XO, Nurieva R, et al. (2009) Critical regulation of early Th17 cell differentiation by interleukin-1 signaling. Immunity 30: 576-587.
- Rider P, Carmi Y, Voronov E, Apte RN (2013) Interleukin-1α Semin. Immunol 25: 430-438.
- Dinarello CA (2009) Immunological and Inflammatory Functions of the Interleukin-1 Family. Annu. Rev. Immunol 27: 519-550.
- Steifer T, Lewandowski M (2019) Ultrasound tissue characterization based on the Lempel-Ziv complexity with application to breast lesion classification, Biomedical Signal Processing and Control 51: 235-242.
- Cheng H D, Shan J, Ju W, Guo Y and Zhang L (2010) Automated breast cancer detection and classification using ultrasound images: a survey, Pattern Recognition 43: 299-317.
- Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft DK 2018, AWMF) www.leitlinienprogrammonkologie.de/leitlinien/mammakarzinom/ (last accessed on 30 April 2019) Berli. Interdisziplinäre S3-Leitlinie für die Früherkennung, Diagnostik, Therapie und Nachsorge des Mammakarzinoms
- Bower JE, Ganz PA, Irwin MR, Castellon S, Arevalo J, et al. (2013) Cytokine genetic variations and fatigue among patients with breast cancer. Journal of Clinical Oncology 31: 1656-1661.
- Walthers EM. Springer. Berlin, Heidelberg: 2016. ACR BI-RADS Atlas der Mammadiagnostik.
- Guray M and Sahin AA (2006) Benign breast diseases: classification, diagnosis, and management. Oncologist 11: 435-439.
- Orta T, Günebakan S (2012) The effect of aging on micronuclei frequency and proliferation in human peripheral blood lymphocytes. Indian j. Hum. Genet 18: 95.
- Emiro?ullar? ON, Tunçay A, ?ener EF, Taheri S, Ünal A, et al. (2018) Investigation of interleukin 1 alpha gene promoter polymorphism in hemodialysis patients with arteriovenous fistula thrombosis. Erciyes Med J 40: 18-22
- Lafrenie R, Bewick M, Buckner C, Conlon M (2023) Plasma Cytokine Levels and Cytokine Genetic Polymorphisms in Patients with Metastatic Breast Cancer Receiving High-Dose Chemotherapy. Immuno 3: 16-34.